- Blog
- Download game inazuma eleven ps2 iso emuparadise
- Night owl x app for pc
- Die antwoord enter the ninja clean version
- Tamil bible audio
- Astro saber dx starbound
- Android jones live visuals
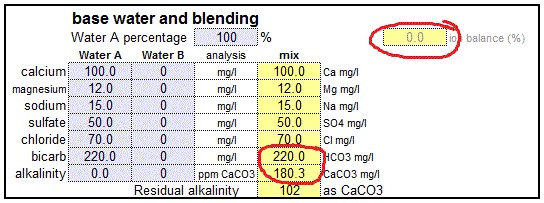
- How to calculate ppm of caco3 in your water samplke
- Visual studio javascript intellisense in html5
- Sindhi koki
- Lil webbie savage life 2 songs
- Planet centauri upgrade gems
- Docker ssh agent 2017
- Adobe acrobat 7-0 professional help
- Brothers in arms pc requirements

Calcium hardness is best monitored with a drop-count titration, as test strips can only measure total hardness.

To avoid damage to pools and spas from unbalanced water, test calcium hardness at least monthly.
As mentioned, the excess calcium carbonate will precipitate as crusty, grayish white scale on surfaces, piping, and equipment. Specifically, water with high calcium hardness gets cloudy unless the alkalinity and/or pH are low enough to compensate. Colored water can be cured with a "metal out" product, or chelating agent, and some of these are reportedly successful at removing certain surface stains. A thin layer of the concrete surface of the pool may dissolve as well, and copper can be lost from piping and/or heat exchangers.ĭamage from corrosive water cannot be reversed, only repaired by resurfacing the concrete and replacing piping. Corrosive water attacks plaster, concrete, grout, and metal, resulting in etching, pitting, and surface stains and/or colored water caused by metal pulled out of piping, fittings, and equipment.Ĭan damage from scaling water be reversed? By reducing the SI to around -1.0 for a short time (usually by lowering pH), some calcium deposits in the filter and circulation piping can be dissolved, and the water's flow may even remove chunks of loosened scale. When the SI is +0.5 or more, the unbalanced water is trending toward scaling, meaning conditions are right for calcium carbonate to come out of solution and deposit on surfaces as "scale." When the SI is -0.3 or less, the unbalanced water is trending toward corrosivity. Its calcium hardness, pH, and total alkalinity are acting in harmony with one another. When the SI value is zero, the water is properly balanced. The value one arrives at reflects the water's degree of saturation with calcium carbonate. All but the mathletes among us use a water balance calculator like Taylor Technologies' Watergram ® to do the number crunching involved. Called the Saturation Index (SI) formula, it considers the interrelationships of four chemical factors-calcium hardness pH total alkalinity, as corrected for the contribution of any cyanuric acid stabilizer in use and, to a much lesser extent, the total dissolved solids level-plus one physical factor, water temperature. While hardness in water consists of both calcium and magnesium salts ("total hardness"), only the calcium component is relevant in the water balance calculation for pools and spas. The Role of Calcium Hardness in Water Balance Current industry standards call for maintaining calcium hardness in the ideal range of 200–400 ppm in pools and 150–250 ppm in spas. Reader note: An edited version of this article originally appeared in the summer 2007 issue of The Edge, a publication of the Northeast Spa & Pool Association.Ĭalcium hardness must be actively managed-along with pH and total alkalinity-to keep water in proper chemical balance.
